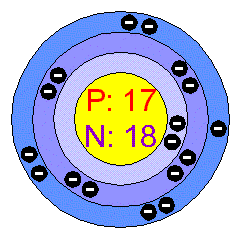
(iv) Carbon anode is used when molten magnesium chloride is electrolyzed. The base value of hydrogen was later increased by 0.10 and caesiums electronegativity was later refined to 0.79 however, no refinements have been made for francium as no experiment has been conducted. Therefore, they have similar chemical properties because chemical properties are dependent on electronic configuration of an element. The electronegativity of francium was chosen by Pauling as 0.7, close to that of caesium (also assessed 0.7 at that point). (iv) As the two atoms in (iii) above are isotopes and hence have same electronic configuration. Hence, its electronic configuration = 2, 8, 7 Sources, facts, uses, scarcity (SRI), podcasts, alchemical symbols, videos and images. What is the ratio of Cl2 to Mg in MgCl2 if you have 0.03mol Mg and 0.06mol Cl2 MgCl2 contain. Element Chlorine (Cl), Group 17, Atomic Number 17, p-block, Mass 35.45. (ii) Atomic number of Chlorine is 17 so it has 17 electrons. You can ignore Cl as the atom with atomic number 18 is argon a noble gas- one higher than Cl is F. 
Positive charges - in the nucleus of it's atomĪn atom is the smallest particle of an element which can take part in a chemical reaction and is considered the basic unit of matter. Chlorine is an element of the XVII group of Mendeleevs periodic table of chemical elements, has an atomic number of 17 and an atomic mass of 35,453.Isotopes are atoms that have the same number of protons but. (i) Atomic number of an element is the number of : In the periodic table of the elements, elements are arranged in order of ascending atomic number.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
